Making Cyanide Work Faster
Overview
The Problem:
How to provide statistically valid evidence of improvements in the gold leaching process brought about by the introduction of a new oxygenation system.
The Data Analysis Australia Approach:
Multiple mathematical models were fitted to the oxygenation system, with automatic significance testing of any differences.
The Result:
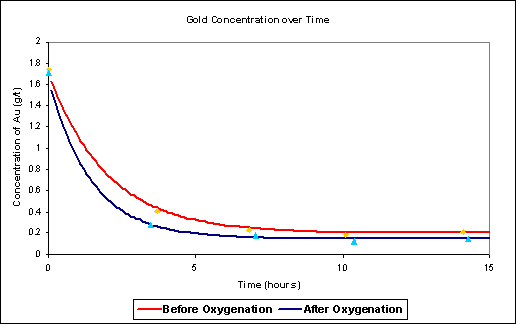
A suite of functions implemented in S-Plus in a user-friendly environment, which provides graphical representations of before and after results and tests for statistically significant differences.
Background
Gold mining is one of Western Australia's largest mineral industries, with an annual production of over 192 tonnes worth around $3.2 billion. Almost all mines use a cyanide leaching process to extract the gold from the ore. In this process the finely milled ore is mixed with a sodium cyanide solution that dissolves the gold. The reaction between the gold and the cyanide ions is described by Elsner's equation:
4Au + 8CN- + O2 + 2H2O = 4Au(CN)2- + 4OH-
Activated carbon is then used to recover the gold from this solution. The leaching takes place in large tanks holding many thousands of cubic metres of solution and generally takes several days.
Naturally all miners want this process to be as efficient as possible and to maximise the gold extracted from the ore. Across Australia over $100 million worth of cyanide is used each year in this process. Hence improving the efficiency is of value not only in terms of the extra gold recovered but also in the reduction of costs. In addition, if the leaching process can be accelerated then a plant can increase its throughput, further reducing the cost of treating each tonne of ore.
The Problem
One method of improving the leach process is to ensure that sufficient oxygen is present. In order to do this, Multi Mix Systems developed a series of injectors that mix controlled amounts of oxygen into the ore slurry. However, it is important to be able to measure the improvements gained so that a true cost benefit analysis can be carried out.
Measuring the improvement is not easy since the ore being fed into a typical processing plant is constantly changing, reflecting the varying gold grade in the mine. This means that simple measures such as the amount of gold recovered can be confusing. Multi Mix Systems realised that it was important to estimate the actual leaching rates, and called upon Data Analysis Australia to assist with this.
It was important that the solution not only be statistical best practice but also that it could be readily implemented by Multi Mix Systems consultants in the field.
The Data Analysis Australia Approach
As a starting point Data Analysis Australia reviewed applicable mathematical models for the leaching process. Since all these models have approximations, the evaluation was based on their ability to fit actual data in a reliable manner. On the basis of this statistical examination, one particular model was chosen as the preferred model for regular analysis.
The next step was the development of a software solution for ongoing use by Multi Mix Systems. Data Analysis Australia selected the statistical system S-Plus for this. S-Plus is sometimes called the "statistician's statistical package" since it includes many advanced features and is often the environment where new statistical procedures are made available. In this case S-Plus could provide the necessary non-linear fitting algorithms as well as the programmability to incorporate the special features of this problem. (The longitudinal structure of some of the data and the variation in the head grades required special attention.)
The Result
The software system developed by Data Analysis Australia is a user friendly environment for assessing the effect of the oxygenation equipment, providing tests of statistical significance and readily interpreted graphical output. It enables the improvements in both leaching rates and recovery rates to be estimated and monitored. Multi Mix Systems now provide this package as a value added service to their clients.
October 1999
